Third paper from a Short-Term Scientific Mission (STSM) (September 12, 2015)
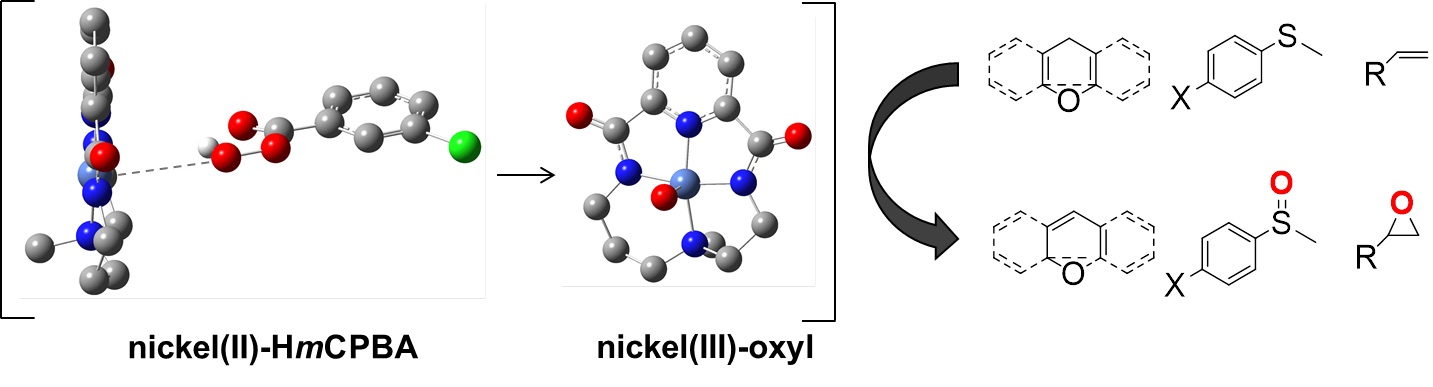
In this work, the group of Dr. Anna Company reports the formation of a highly reactive nickel-oxygen species that has been trapped following reaction of a NiII precursor bearing a macrocyclic bis(amidate) ligand with meta-chloroperbenzoic acid (HmCPBA).
This compound is only detectable at temperatures below 250K and is much more reactive toward organic substrates (i.e., C-H bonds, C=C bonds, and sulfides) than previously reported well-defined nickel-oxygen species.
Remarkably, this species is formed by heterolytic O-O bond cleavage of a Ni-HmCPBA precursor, which is concluded from experimental and computational data.
On the basis of spectroscopy and DFT calculations, this reactive species is proposed to be a NiIII-oxyl compound.
Several spectroscopic techniques and theoretical methods were necessary in order to establish the nature of this nickel-oxygen species. XAS experiments were carried out by Dr. Vlad Martin-Diaconescu (University of Girona), rRaman by Prof. Wesley Browne (University of Groningen), DFT calculations by Dr. Julio Lloret-Fillol (ICIQ Tarragona) and EPR spectroscoy by Dr. Kallol Ray (Humboldt Universität zu Berlin). In particular, these studies were performed during a one week STSM hosted by Kallol Ray by Teresa Corona who is a PhD student of Anna Company. This work is now accepted for publication in Chemistry - A European Journal:
"Reactivity of a Nickel(II) Bis(amidate) Complex with meta-Chloroperbenzoic Acid: Formation of a Potent Oxidizing Species"
Teresa Corona, Florian F. Pfaff, Ferran Acuña-Parés, Dr. Apparao Draksharapu, Dr. Christopher J. Whiteoak, Dr. Vlad Martin-Diaconescu, Dr. Julio Lloret-Fillol, Prof. Dr. Wesley R. Browne, Dr. Kallol Ray and Dr. Anna Company*
Chem. Eur. J. 2015, 21, 15029-15038
DOI: 10.1002/chem.201501841