Sixteenth paper from a Short-Term Scientific Mission (STSM) (September 11, 2017)
The distribution of metal ions over different sites is a fundamental issue in coordination chemistry. The balance between normal and inverse spinels has become a textbook example of the importance of ligand field stabilization energy (LFSE). Related issues are less well explored in the context of molecular clusters. This is of profound importance in biological chemistry because heteronuclear clusters are found in the reaction centers of many important enzymes. In the present manuscript the groups of C. P. Raptopoulou, Y. Sanakis (Institute of Nanoscience and Nanotechnology, NCSR Demokritos, Greece) and J. E. McGrady (Department of Chemistry, University of Oxford, UK) address the site preference in hetero-metallic polynuclear transition metal clusters.
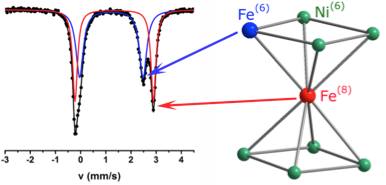
The synthesis and characterization of a family of hetero-metallic enneanuclear clusters with general formula
[Fe9-xNixμ4-OH)2(O2CMe)8(py2CO2)4] are described.
All clusters contain one 8-coordinate (rarely encountered in Fe(II) and Ni(II) chemistry) and eight 6-coordinate metal sites. The distribution of Fe(II) and Ni(II) over the two distinct metal sites and the formulation of the clusters are established via crystallography and spectroscopy. A notable site preference for iron to occupy the 8-coordinate site in low Fe-content species is observed via Mössbauer spectroscopy. Density functional theory further corroborated that Ni(II) ions preferably occupy the octahedral sites, hence leaving the 8-coordinate site for the Fe(II) ion. The work was supported by an STSM mission (COST-STSM-CM1305-34875 ) by Karrar Al-Ameed, a PhD student at the Oxford University to the group of Yiannis Sanakis, NCSR Demokritos. The results of this collaboration were recently published in Dalton Transactions.